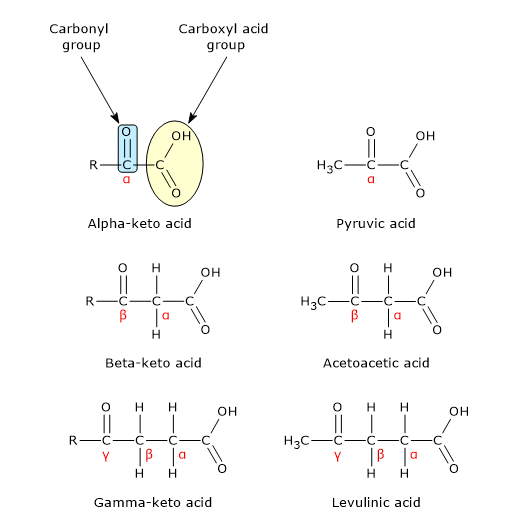
Keto acids or ketoacids are organic compounds containing two functional groups: a carboxyl acid group (−COOH) and a carbonyl group (˂C=O).
Based on the position of the carbonyl group relative to the carboxylic acid group, to which IUPAC nomenclature rules assign the highest priority, ketoacids are classified as α-keto acids, β-keto acids, and γ-keto acids.[1]
Keto acids, and in particular α-keto acids, are very important in biochemistry, being involved in many metabolic pathways.[2]
In Summary: Key Points
- Definition and structure: keto acids are organic compounds containing both a carboxylic acid group and a carbonyl group within their molecular structure.
- IUPAC classification: they are classified into alpha (α), beta (β), and gamma (γ) keto acids based on the position of the carbonyl group relative to the carboxylic acid.
- Role of alpha-keto acids: pyruvic acid (the end product of glycolysis), oxaloacetic acid, and α-ketoglutaric acid are vital metabolic intermediates driving the citric acid cycle, gluconeogenesis, and amino acid metabolism.
- Significance of beta and gamma: beta-ketoacids (such as acetoacetic acid) are key to ketone body production and gut microbiota pathways, while gamma-keto acids (such as levulinic acid) originate from cellulose catabolism.
α-Keto acids
They have the carbonyl group adjacent to the carboxylic acid. Many of these compounds, in the form of their conjugate bases, have important biological functions. Below are some examples.[2]
Pyruvic acid, the simplest α-keto acid, is the end metabolic product of glycolysis.
Oxaloacetic acid and α-ketoglutaric acid are intermediates of the citric acid cycle.
α-Ketoacids can arise from transamination and oxidative deamination reactions of amino acids. In transamination reactions, the α amino group of the amino acid is transferred to an α-keto acid, usually α-ketoglutarate, with the formation of a new amino acid and a new α-keto acid. These reactions are catalyzed by enzymes called aminotransferases or transaminases (EC 2.6.1.-).
α-Keto acid + Amino acid ⇄ New amino acid + New α-keto acid
In oxidative deaminations, amino acids are converted into the corresponding α-keto acids by removing the amino group, which is converted to ammonia and replaced by a carbonyl group. Since the reaction is reversible, ketoacids are also precursors of amino acids.
Note: ammonia is a toxic compound, and is converted into the safer compound urea via the urea cycle in the liver.[2]
Pyruvate, oxaloacetate and α-ketoglutarate, the latter via oxaloacetate, are the entry points into gluconeogenesis of the carbon skeleton of many glucogenic amino acids.[3]
It has also been observed that, in vitro, murine and human tumor cell lines secrete 2-keto acids into the tumor microenvironment, such as α-ketoisocaproate, α-keto-β-methylvalerate and α-ketoisovalerate, which are capable to influencing the anti-tumor activity of macrophages.[4]
β-Keto acids
They have the carbonyl group at the second carbon from the carboxylic acid.
Examples of β-ketoacids are acetoacetic acid, the simplest one, and β-hydroxybutyric acid, which are two of the three ketone bodies, together with acetone, produced by the hepatocyte when acetyl-CoA is produced in excess of the capacity of citrate synthase (EC 2.3.3.1), namely, of citric acid cycle to oxidize it fully, as during prolonged fasting or diets very low in carbohydrates.
Note that acetoacetyl-CoA and β-hydroxybutyryl-CoA, namely, the activated forms of these β-keto acids, are also intermediates in the butyric acid synthesis pathway which occurs in most butyrate-producing bacteria of the gut microbiota.[5][6][7]
γ-Keto acids
They have the carbonyl group at the third carbon from the carboxylic acid.
An example is levulinic acid, the simplest one, which arises from the catabolism of cellulose.
References
- ^ IUPAC, Pure Appl Chem. https://iupac.org/
- ^ a b c Nelson D.L., Cox M.M. Lehninger. Principles of biochemistry. 8th Edition. W.H. Freeman and Company, 2021.
- ^ D’Andrea G. Classifying amino acids as gluco(glyco)genic, ketogenic, or both. Biochem Educ 2000;28(1):27-28. doi:10.1016/s0307-4412(98)00271-4
- ^ Cai Z., Li W., Brenner M., Bahiraii S., Heiss E.H., Weckwerth W. Branched-chain ketoacids derived from cancer cells modulate macrophage polarization and metabolic reprogramming. Front Immunol 2022;13:966158. doi:10.3389/fimmu.2022.966158.
- ^ Miller T.L., Wolin M.J. Pathways of acetate, propionate, and butyrate formation by the human fecal microbial flora. Appl Environ Microbiol 1996;62(5):1589-92. doi:10.1128/aem.62.5.1589-1592
- ^ Portincasa P., Bonfrate L.,Vacca M., De Angelis M., Farella I., Lanza E., Khalil M.,Wang D.Q.-H., Sperandio M., Di Ciaula A. Gut microbiota and short chain fatty acids: implications in glucose homeostasis. Int J Mol Sci 2022;23:1105. doi:10.3390/ijms23031105
- ^ Pryde S.E., Duncan S.H., Hold G.L., Stewart C.S., Flint H.J. The microbiology of butyrate formation in the human colon. FEMS Microbiol Lett 2002;217(2):133-9. doi:10.1111/j.1574-6968.2002.tb11467.x
Domande Frequenti
What are keto acids and how are they classified according to IUPAC nomenclature?
Keto acids are organic compounds containing a carboxylic acid group and a carbonyl group. Based on the position of the carbonyl group relative to the carboxyl group, IUPAC rules classify them into alpha (α), beta (β), and gamma (γ) keto acids.
What is the biological importance of alpha-keto acids?
Alpha-keto acids are vital in biochemistry. Pyruvic acid is the metabolic end product of glycolysis, while oxaloacetic acid and α-ketoglutaric acid are key intermediates in the citric acid cycle and entry points for gluconeogenesis.
How are beta-keto acids related to ketone body production?
Beta-keto acids, such as acetoacetic acid, are key ketone bodies produced by the liver when acetyl-CoA exceeds capacity, which typically happens during prolonged fasting or very low-carbohydrate diets.
What is levulinic acid and which group does it belong to?
Levulinic acid is the simplest example of a γ-keto acid. It contains a carbonyl group on the third carbon atom relative to the carboxylic acid group and is produced during the catabolism of the polysaccharide cellulose.