Flavonols are polyphenols belonging to the flavonoid family.
They are colorless molecules that accumulate mainly in the outer and aerial tissues, therefore skin and leaves, of fruit and vegetables, since their biosynthesis is stimulated by light, whereas they are virtually absent in the flesh.
Flavonols are the most common flavonoids in fruit and vegetables, where they are generally present in relatively low concentrations.
Due to their widespread in nature and human diet, they should be taken into consideration when the positive effect on health associated with fruit and vegetable consumption is examined.
Their effect is probably related to their ability to:
- act as antioxidants;
- act as anti-inflammatory agents;
- act as anticancer factors;
- regulate different cellular signaling pathways; an example is the action of quercetin, the most widespread flavonols, on the oxidative stress-induced MAPK activities.
Contents
Chemical structure
Chemically, these molecules differ from many other flavonoids since they have a double bond between positions 2 and 3 and an oxygen (a ketone group) in position 4 of the C ring, like flavones from which, however, they differ in the presence of a hydroxyl group at the position 3. Therefore, flavonol skeleton is a 3-hydroxyflavone.
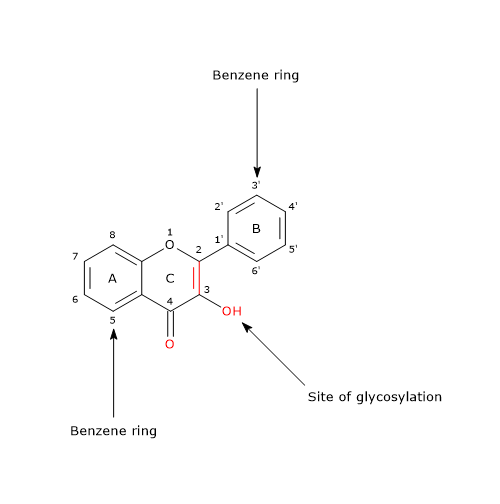
The 3-hydroxyl group can link a sugar, that is, it can be glycosylated.
Like many other flavonoids, most of them is found in fruit and vegetables, and in plant-derived foods, in glycosylated form. The sugar associated with flavonols is often glucose or rhamnose, but other sugars may also be involved, such as:
- galactose;
- arabinose;
- xylose;
- glucuronic acid.
Flavonols are mainly represented by glycosides of:
- quercetin;
- kaempferol;
- myricetin;
- isorhamnetin.
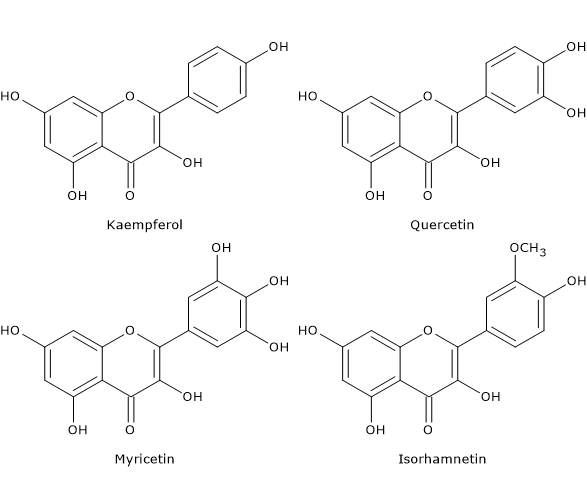
The most ubiquitous compounds are glycosylated derivatives of quercetin and kaempferol; in nature, these two molecules have respectively about 280 and 350 different glycosidic combinations.
Finally, it should be underlined that sugar moiety influences flavonol bioavailability.
Food sources
The major sources in human diet are:
- fruit;
- vegetables;
- beverages such as red wine and tea.
In human diet, the richest source are capers, which contain up to 490 mg/100 g fresh weight (FW), but they are also abundant in onions, leeks, broccoli, curly kale, berries, for example blueberries, grapes and some herbs and spices, for example dill weed (Anethum graveolens). In these sources, their content ranges between 10 and 100 mg/100 g FW.
Even cocoa, green tea, black tea, and red wine are good sources of flavonols. In wine, together with other polyphenols such as catechins, proanthocyanidins and low molecular weight polyphenols, they contribute to the astringency of the beverage.
Main flavonols in foods
The main flavonols in foods, listed in decreasing order of abundance, are quercetin, kaempferol, myricetin and ishoramnetin.
Quercetin
The richest sources of quercetin are capers, followed by onions, asparagus, lettuce and berries; in many other fruit and vegetables, it is present in smaller amounts, between 0.1 and 5 mg/100 g FW.
This flavonol is also present in cocoa and it could be one of its main protective agents against LDL oxidation.
Together with isoflavones, quercetin glycosides are the most well-absorbed polyphenols, followed by flavanones and catechins. Convesely, gallic acid derivatives of catechins are among the least well absorbed polyphenols, together with anthocyanins and proanthocyanidins.
Kaempferol
Typical dietary sources of kaempferol include vegetables, such as spinach, kale and endive, with concentrations between 0.1 and 27 mg/100 g FW, and some spices such as chives, fennel and tarragon, with concentrations between 6.5 and 19 mg/100 g FW.
Fruit is a poor source of the molecule, with content down to 0.1 mg/100 g FW.
Myricetin
Myricetin is the third most abundant flavonol. It is found in some spices, such as oregano, parsley, and fennel, with concentrations between 2 and 20 mg/100 g FW, but also in tea, 0.5-1.6 mg/100 ml, and red wine, 0-9.7 mg/100 ml.
In fruit, it is only found in high concentrations in berries, while in most other fruit and vegetables it is present in a content of less than 0.2 mg/100 g FW.
Isorhamnetin
A fourth flavonol, less abundant than the previous ones, is isorhamnetin. It is only present in some foods such as some spices: chives, 5.0-8.5 mg/100 g FW, fennel, 9.3 mg/100 g FW, tarragon, 5 mg/100 g FW.
In fruit and vegetables it is only present in almonds, with a concentration between 1.2 and 10.3 mg/100 g FW, pears and onions.
References
- de la Rosa L.A., Alvarez-Parrilla E., Gonzàlez-Aguilar G.A. Fruit and vegetable phytochemicals: chemistry, nutritional value, and stability. 1th Edition. Wiley J. & Sons, Inc., Publication, 2010
- Han X., Shen T. and Lou H. Dietary polyphenols and their biological significance. Int J Mol Sci 2007;9:950-988. doi:10.3390/i8090950
- Manach C., Scalbert A., Morand C., Rémésy C., and Jime´nez L. Polyphenols: food sources and bioavailability. Am J Clin Nutr 2004;79(5):727-747. doi:10.1093/ajcn/79.5.727
- Tsao R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010;2:1231-1246. doi:10.3390/nu2121231