Gluconeogenesis is a metabolic pathway that leads to the synthesis of glucose from pyruvate and other non-carbohydrate precursors, even in non-photosynthetic organisms.[1][6]
It occurs in all microorganisms, fungi, plants and animals, and the reactions are essentially the same, leading to the synthesis of one glucose molecule from two pyruvate molecules. Therefore, it is in essence glycolysis in reverse, which instead goes from glucose to pyruvate, and shares seven enzymes with it.[5]
Glycogenolysis is quite distinct from gluconeogenesis: it does not lead to de novo production of glucose from non-carbohydrate precursors, as shown by its overall reaction:
Glycogen or (glucose)n → n glucose molecules
The following discussion will focus on gluconeogenesis that occurs in higher animals, and in particular in the liver of mammals.
Contents
- Why is it important?
- Where does it occur?
- Irreversible steps
- From pyruvate to phosphoenolpyruvate
- From fructose 1,6-bisphosphate to fructose 6-phosphate
- From glucose 6-phosphate to glucose
- Gluconeogenesis: energetically expensive
- Coordinated regulation of gluconeogenesis and glycolysis
- Regulation of gluconeogenesis
- Precursors
- References
Why is it important?
Gluconeogenesis is an essential metabolic pathway for at least two reasons.
- It ensures the maintenance of appropriate blood glucose levels when the liver glycogen is almost depleted and no carbohydrates are ingested.[6]
- Maintaining blood glucose within the normal range, 3.3 to 5.5 mmol/L (60 and 99 mg/dL), is essential because many cells and tissues depend, largely or entirely, on glucose to meet their ATP demands; examples are red blood cells, neurons, skeletal muscle working under low oxygen conditions, the medulla of the kidney, the testes, the lens and the cornea of the eye, and embryonic tissues. For example, glucose requirement of the brain is about 120 g/die that is equal to:
over 50 percent of the total body stores of the monosaccharide, about 210 g, of which 190 g are stored as liver and muscle glycogen, and 20 g are found in free form in body fluids;
about 75 percent of the daily glucose requirement, about 160 g.[1]
During fasting, as in between meals or overnight, the blood glucose levels are maintained within the normal range due to hepatic glycogenolysis, and to the release of fatty acids from adipose tissue and ketone bodies by the liver. Fatty acids and ketone bodies are preferably used by skeletal muscle, thus sparing glucose for cells and tissues that depend on it, primarily red blood cells and neurons. However, after about 18 hours of fasting or during intense and prolonged exercise, glycogen stores are depleted and may become insufficient. At that point, if no carbohydrates are ingested, gluconeogenesis becomes important.
And, the importance of gluconeogenesis is further emphasized by the fact that if the blood glucose levels fall below 2 mmol/L, unconsciousness occurs.
- The excretion of pyruvate would lead to the loss of the ability to produce ATP through aerobic respiration, i.e. more than 10 molecules of ATP for each molecule of pyruvate oxidized.
Where does it occur?
In higher animals, gluconeogenesis occurs in the liver, kidney cortex and epithelial cells of the small intestine, that is, the enterocytes.[5]
Quantitatively, the liver is the major site of gluconeogenesis, accounting for about 90 percent of the synthesized glucose, followed by kidney cortex, with about 10 percent.[1] The key role of the liver is due to its size; in fact, on a wet weight basis, the kidney cortex produces more glucose than the liver.
In the kidney cortex, gluconeogenesis occurs in the cells of the proximal tubule, the part of the nephron immediately following the glomerulus. Much of the glucose produced in the kidney is used by the renal medulla, while the role of the kidney in maintaining blood glucose levels becomes more important during prolonged fasting and liver failure. It should, however, be emphasized that the kidney has no significant glycogen stores, unlike the liver, and contributes to maintaining blood glucose homeostasis only through gluconeogenesis and not through glycogenolysis.
Part of the gluconeogenesis pathway also occurs in the skeletal muscle, cardiac muscle, and brain, although at very low rate.
In adults, muscle is about 18 the weight of the liver; therefore, its de novo synthesis of glucose might have quantitative importance. However, the release of glucose into the circulation does not occur because these tissues, unlike liver, kidney cortex, and enterocytes, lack glucose 6-phosphatase (EC 3.1.3.9), the enzyme that catalyzes the last step of gluconeogenesis.[6] Therefore, the production of glucose 6-phosphate, including that from glycogenolysis, does not contribute to the maintenance of blood glucose levels, and only helps to restore glycogen stores, in the brain small and limited mostly to astrocytes. For these tissues, in particular for skeletal muscle due to its large mass, the contribution to blood glucose homeostasis results only from the small amount of glucose released in the reaction catalyzed by enzyme debranching (EC 3.2.1.33) of glycogenolysis.
With regard to the cellular localization, most of the reactions occur in the cytosol, some in the mitochondria, and the final step) within the endoplasmic reticulum cisternae.
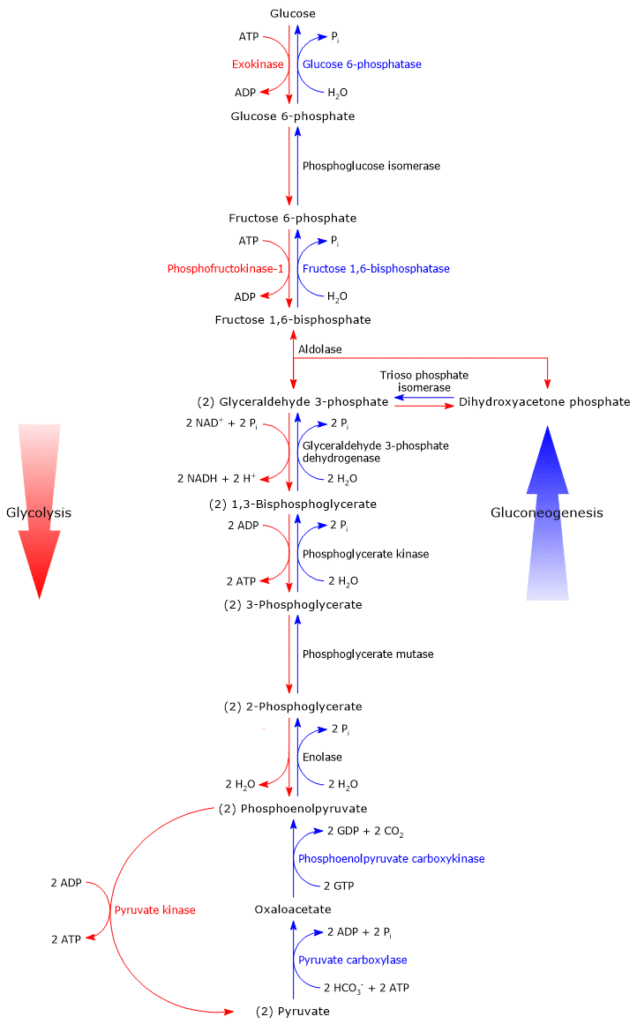
Irreversible steps
As previously said, gluconeogenesis is in essence glycolysis in reverse. And, of the ten reactions that constitute gluconeogenesis, seven are shared with glycolysis; these reactions have a ΔG close to zero, therefore easily reversible. However, under intracellular conditions, the overall ΔG of glycolysis is about -63 kJ/mol (-15 kcal/mol) and of gluconeogenesis about -16 kJ/mol (-3.83 kcal/mol), namely, both the pathways are irreversible.[5]
The irreversibility of the glycolytic pathway is due to three strongly exergonic reactions, that cannot be used in gluconeogenesis, and listed below.
- The phosphorylation of glucose to glucose 6-phosphate, catalyzed by hexokinase (EC 2.7.1.1) or glucokinase (EC 2.7.1.2).
ΔG = -33.4 kJ/mol (-8 kcal/mol)
ΔG°’ = -16.7 kJ/mol (-4 kcal/mol) - The phosphorylation of fructose 6-phosphate to fructose 1,6-bisphosphate, catalyzed by phosphofructokinase-1 or PFK-1 (EC 2.7.1.11)
ΔG = -22.2 kJ/mol (-5.3 kcal/mol)
ΔG°’ = -14.2 kJ/mol (-3.4 kcal/mol) - The conversion of phosphoenolpyruvate or PEP to pyruvate, catalyzed by pyruvate kinase (EC 2.7.1.40)
ΔG = -16.7 kJ/mol (-4.0 kcal/mol)
ΔG°’ = -31.4 kJ/mole (-7.5 kcal/mol)
In gluconeogenesis, these three steps are bypassed by enzymes that catalyze irreversible steps in the direction of glucose synthesis.[6] This ensures the irreversibility of the metabolic pathway.
Below, such reactions are analyzed.
From pyruvate to phosphoenolpyruvate
The first step of gluconeogenesis that bypasses an irreversible step of glycolysis, namely the reaction catalyzed by pyruvate kinase, is the conversion of pyruvate to phosphoenolpyruvate.[8]
Phosphoenolpyruvate is synthesized through two reactions catalyzed, in order, by the enzymes:
- pyruvate carboxylase (EC 6.4.1.1);
- phosphoenolpyruvate carboxykinase or PEP carboxykinase (EC 4.1.1.32).
Pyruvate → Oxaloacetate → Phosphoenolpyruvate
Pyruvate carboxylase catalyzes the carboxylation of pyruvate to oxaloacetate, with the consumption of one ATP. The enzyme requires the presence of magnesium or manganese ions
Pyruvate + HCO3–+ ATP → Oxaloacetate + ADP + Pi
The enzyme, discovered in 1960 by Merton Utter, is a mitochondrial protein composed of four identical subunits, each with catalytic activity. The subunits contain a biotin prosthetic group, covalently linked by amide bond to the ε-amino group of a lysine residue, that acts as a carrier of activated CO2 during the reaction. An allosteric binding site for acetyl-CoA is also present in each subunit.
It should be noted that the reaction catalyzed by pyruvate carboxylase, leading to the production of oxaloacetate, also provides intermediates for the citric acid cycle or Krebs cycle.
Phosphoenolpyruvate carboxykinase is present, approximately in the same amount, in mitochondria and cytosol of hepatocytes. The isoenzymes are encoded by separate nuclear genes.
The enzyme catalyzes the decarboxylation and phosphorylation of oxaloacetate to phosphoenolpyruvate, in a reaction in which GTP acts as a donor of high-energy phosphate. PEP carboxykinase requires the presence of both magnesium and manganese ions. The reaction is reversible under normal cellular conditions.
Oxaloacetate + GTP ⇄ PEP + CO2 + GDP
During this reaction, a CO2 molecule, the same molecule that is added to pyruvate in the reaction catalyzed by pyruvate carboxylase, is removed. Carboxylation-decarboxylation sequence is used to activate pyruvate, since decarboxylation of oxaloacetate facilitates, makes thermodynamically feasible, the formation of phosphoenolpyruvate.
More generally, carboxylation-decarboxylation sequence promotes reactions that would otherwise be strongly endergonic, and also occurs in the citric acid cycle, in the pentose phosphate pathway, also called the hexose monophosphate pathway, and in the synthesis of fatty acids.
The levels of PEP carboxykinase before birth are very low, while its activity increases several fold a few hours after delivery. This is the reason why gluconeogenesis is activated after birth.
The sum of the reactions catalyzed by pyruvate carboxylase and phosphoenolpyruvate carboxykinase is:
Pyruvate + ATP + GTP + HCO3– → PEP + ADP + GDP + Pi + CO2
ΔG°’ of the reaction is equal to 0.9 kJ/mol (0.2 kcal/mol), while standard free energy change associated with the formation of pyruvate from phosphoenolpyruvate by reversal of the pyruvate kinase reaction is + 31.4 kJ/mol (7.5 kcal/mol).
Although the ΔG°’ of the two steps leading to the formation of PEP from pyruvate is slightly positive, the actual free-energy change (ΔG), calculated from intracellular concentrations of the intermediates, is very negative, -25 kJ/mol (-6 kcal/mol). This is due to the fast consumption of phosphoenolpyruvate in other reactions, that maintains its concentration at very low levels. Therefore, under cellular conditions, the synthesis of PEP from pyruvate is irreversible.[5]
It is noteworthy that the metabolic pathway for the formation of phosphoenolpyruvate from pyruvate depends on the precursor: pyruvate or alanine, or lactate.
Phosphoenolpyruvate precursors: pyruvate or alanine
The bypass reactions described below predominate when alanine or pyruvate is the glucogenic precursor.
Pyruvate carboxylase is a mitochondrial enzyme, therefore pyruvate must be transported from the cytosol into the mitochondrial matrix. This is mediated by transporters located in the inner mitochondrial membrane, referred to as MPC1 and MPC2. These proteins, associating, form a hetero-oligomer that facilitates pyruvate transport.[4]
Pyruvate can also be produced from alanine in the mitochondrial matrix by transamination, in the reaction catalyzed by alanine aminotransferase (EC 2.6.1.2), while the amino group is then converted into urea through the urea cycle.
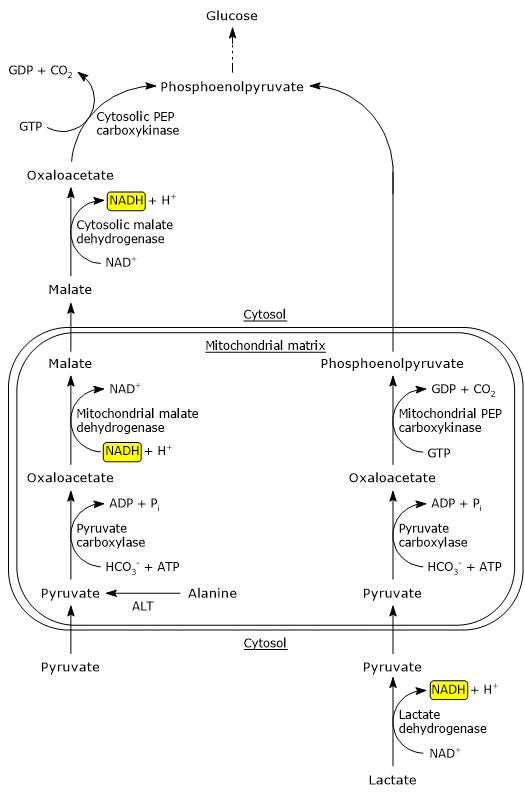
Since the enzymes involved in the later steps of gluconeogenesis, except glucose-6-phosphatase, are cytosolic, the oxaloacetate produced in the mitochondrial matrix is transported into the cytosol. However, there are no oxaloacetate transporters in the inner mitochondrial membrane. The transfer to the cytosol occurs as a result of its reduction to malate, that, on the contrary, can cross the inner mitochondrial membrane. The reaction is catalyzed by mitochondrial malate dehydrogenase (EC 1.1.1.37), an enzyme also involved in the citric acid cycle, where the reaction proceeds in the reverse direction. In the reaction NADH is oxidized to NAD+.
Oxaloacetate + NADH + H+ ⇄ Malate + NAD+
Although ΔG°’ of the reaction is highly positive, under physiological conditions, ΔG is close to zero, and the reaction is easily reversible.
Malate crosses the inner mitochondrial membrane through a component of the malate-aspartate shuttle, the malate-α-ketoglutarate transporter. Once in the cytosol, the malate is re-oxidized to oxaloacetate in the reaction catalyzed by cytosolic malate dehydrogenase. In this reaction NAD+ is reduced to NADH.
Malate + NAD+ → Oxaloacetate + NADH + H+
Note: Malate-aspartate shuttle is the most active shuttle for the transport of NADH-reducing equivalents from the cytosol into the mitochondria. It is found in mitochondria of liver, kidney, and heart.
The reaction enables the transport into the cytosol of mitochondrial reducing equivalents in the form of NADH.[8] This transfer is needed for gluconeogenesis to proceed, as in the cytosolic the NADH, oxidized in the reaction catalyzed by glyceraldehydes 3-phosphate dehydrogenase (EC 1.2.1.12), is present in very low concentration, with a [NADH]/[NAD+] ratio equal to 8×10-4, about 100,000 times lower than that observed in the mitochondria.[5]
Finally, the oxaloacetate is converted to phosphoenolpyruvate in the reaction catalyzed by PEP carboxykinase.
Phosphoenolpyruvate precursor: lactate
Lactate is one of the major gluconeogenic precursors. It is produced for example by:
- red blood cells, that are completely dependent on anaerobic glycolysis for ATP production;
- skeletal muscle during intense exercise, that is, under low oxygen condition, when the rate of glycolysis exceeds the rate of the citric acid cycle and oxidative phosphorylation.[5]
When lactate is the gluconeogenic precursor, PEP synthesis occurs through a different pathway than that previously seen. In the hepatocyte cytosol NAD+ concentration is high and the lactate is oxidized to pyruvate in the reaction catalyzed by the liver isoenzyme of lactate dehydrogenase (EC 1.1.1.27). In the reaction NAD+ is reduced to NADH.
Lactate + NAD+ → Pyruvate + NADH + H+
The production of cytosolic NADH makes unnecessary the export of reducing equivalents from the mitochondria.
Pyruvate enters the mitochondrial matrix to be converted to oxaloacetate in the reaction catalyzed by pyruvate carboxylase. In the mitochondria, oxaloacetate is converted to phosphoenolpyruvate in the reaction catalyzed by mitochondrial pyruvate carboxylase. Phosphoenolpyruvate exits the mitochondria through an anion transporter located in the inner mitochondrial membrane, and, once in the cytosol, continues in the gluconeogenesis pathway.
Note: the synthesis of glucose from lactate may be considered as the part of the Cori cycle that takes place in the liver.
From fructose 1,6-bisphosphate to fructose 6-phosphate
The second step of gluconeogenesis that bypasses an irreversible step of the glycolytic pathway, namely the reaction catalyzed by PFK-1, is the dephosphorylation of fructose 1,6-bisphosphate to fructose 6-phosphate.
This reaction, catalyzed by fructose 1,6-bisphosphatase or FBPasi-1 (EC 3.1.3.11), a Mg2+ dependent enzyme located in the cytosol, leads to the hydrolysis of the C-1 phosphate of fructose 1,6-bisphosphate, without production of ATP.
Fructose 1,6-bisphosphate + H2O → Fructose 6-phosphate + Pi
The ΔG°’ of the reaction is -16.3 kJ/mol (-3.9 kcal/mol), therefore an irreversible reaction.
From glucose 6-phosphate to glucose
The third step of gluconeogenesis that bypasses an irreversible step of the glycolytic pathway, namely the reaction catalyzed by hexokinase or glucokinase, is the dephosphorylation of glucose 6-phosphate to glucose.
This reaction is catalyzed by the catalytic subunit of glucose 6-phosphatase, a protein complex located in the membrane of the endoplasmic reticulum of hepatocytes, enterocytes and cells of the proximal tubule of the kidney.[8] Glucose 6-phosphatase complex is composed of a glucose 6-phosphatase catalytic subunit and a glucose 6-phosphate transporter called glucose 6-phosphate translocase or T1.
Glucose 6-phosphatase catalytic subunit has the active site on the luminal side of the organelle. This means that the enzyme catalyzes the release of glucose not in the cytosol but in the lumen of the endoplasmic reticulum.
Glucose 6-phosphate, both resulting from gluconeogenesis, produced in the reaction catalyzed by glucose 6-phosphate isomerase or phosphoglucose isomerase (EC 5.3.1.9), and glycogenolysis, produced in the reaction catalyzed by phosphoglucomutase (EC 5.4.2.2), is located in the cytosol, and must enter the lumen of the endoplasmic reticulum to be dephosphorylated. Its transport is mediated by glucose-6-phosphate translocase.
The catalytic subunit of glucose 6-phosphatase, a Mg2+-dependent enzyme, catalyzes the last step of both gluconeogenesis and glycogenolysis. And, like the reaction catalyzed by fructose 1,6-bisphosphatase, this reaction leads to the hydrolysis of a phosphate ester.
Glucose 6-phosphate + H2O → Glucose + Pi
It should also be underlined that, due to orientation of the active site, the cell separates this enzymatic activity from the cytosol, thus avoiding that glycolysis, that occurs in the cytosol, is aborted by enzyme action on glucose 6-phosphate.
The ΔG°’ of the reaction is -13.8 kJ/mol (-3.3 kcal/mol), therefore it is an irreversible reaction. If instead the reaction were that catalyzed by hexokinase/glucokinase in reverse, it would require the transfer of a phosphate group from glucose 6-phosphate to ADP. Such a reaction would have a ΔG equal to +33.4 kJ/mol (+8 kcal/mol), and then strongly endergonic. Similar considerations can be made for the reaction catalyzed by FBPase-1.
Glucose and Pi group seem to be transported into the cytosol via different transporters, referred to as T2 and T3, the last one an anion transporter.
Finally, glucose leaves the hepatocyte via the membrane transporter GLUT2, enters the bloodstream and is transported to tissues that require it. Conversely, under physiological conditions, as previously said, glucose produced by the kidney is mainly used by the medulla of the kidney itself.
Gluconeogenesis: energetically expensive
Like glycolysis, much of the energy consumed is used in the irreversible steps of the process.[5]
Six high-energy phosphate bonds are consumed: two from GTP and four from ATP. Furthermore, two molecules of NADH are required for the reduction of two molecules of 1,3-bisphosphoglycerate in the reaction catalyzed by glyceraldehyde 3-phosphate dehydrogenase. The oxidation of NADH causes the lack of production of 5 molecules of ATP that are synthesized when the electrons of the reduced coenzyme are used in oxidative phosphorylation.
Also these energetic considerations show that gluconeogenesis is not simply glycolysis in reverse, in which case it would require the consumption of two molecules of ATP, as shown by the overall glycolytic equation.
Glucose + 2 ADP + 2 Pi + 2 NAD+ → 2 Pyruvate + 2 ATP + 2 NADH + 2 H+ + 2 H2O
Below, the overall equation for gluconeogenesis:
2 Pyruvate + 4 ATP + 2 GTP + 2 NADH+ + 2 H+ + 4 H2O → Glucose + 4 ADP + 2 GDP + 6 Pi + 2 NAD+
At least in the liver, ATP needed for gluconeogenesis derives mostly from the oxidation of fatty acids or of the carbon skeletons of the amino acids, depending on the available “fuel”.
Coordinated regulation of gluconeogenesis and glycolysis
If glycolysis and gluconeogenesis were active simultaneously at a high rate in the same cell, the only products would be ATP consumption and heat production, in particular at the irreversible steps of the two pathways, and nothing more.
For example, considering PFK-1 and FBPasi-1:
ATP + Fructose 6-phosphate → ADP + Fructose 1,6-bisphosphate
Fructose 1,6-bisphosphate + H2O → Fructose 6-phosphate + Pi
The sum of the two reactions is:
ATP + H2O → ADP + Pi + Heat
Two reactions that run simultaneously in opposite directions result in a futile cycle or substrate cycle.
The modulation of the activity of involved enzymes occurs through:
- allosterical mechanisms;
- covalent modifications, such as phosphorylation and dephosphorylation;
- changes in the concentration of the involved enzymes, due to changes in their synthesis/degradation ratio.
Allosteric mechanisms are very rapid and instantly reversible, taking place in milliseconds. The others, triggered by signals from outside the cell, such as hormones, like insulin, glucagon, or epinephrine, take place on a time scale of seconds or minutes, and, for changes in enzyme concentration, hours.
This allows a coordinated regulation of the two pathways, ensuring that when pyruvate enters gluconeogenesis, the flux of glucose through the glycolytic pathway slows down, and vice versa.
Regulation of gluconeogenesis
The regulation of gluconeogenesis and glycolysis involves the enzymes unique to each pathway, and not the common ones.[1]
While the major control points of glycolysis are the reactions catalyzed by PFK-1 and pyruvate kinase, the major control points of gluconeogenesis are the reactions catalyzed by fructose 1,6-bisphosphatase and pyruvate carboxylase.
The other two enzymes unique to gluconeogenesis, glucose-6-phosphatase and PEP carboxykinase, are regulated at transcriptional level.
Pyruvate carboxylase
In the mitochondrion, pyruvate can be converted to:
- acetyl-CoA, in the reactions catalyzed by pyruvate dehydrogenase complex, reaction that connects glycolysis to the Krebs cycle;
- oxaloacetate, in the reaction catalyzed by pyruvate carboxylase, to continue in the gluconeogenesis pathway.
The metabolic fate of pyruvate depends on the availability of acetyl-CoA, that is, by the availability of fatty acids in the mitochondrion.
When fatty acids are available, their beta-oxidation leads to the production of acetyl-CoA, that enters the Krebs cycle and leads to the production of GTP and NADH. When the energy needs of the cell are met, oxidative phosphorylation slows down, the [NADH]/[NAD+] ratio increases, NADH inhibits the citric acid cycle, and acetyl-CoA accumulates in the mitochondrial matrix.[1][5]
Acetyl-CoA is a positive allosteric effector of pyruvate carboxylase, and a negative allosteric effector of pyruvate kinase. Moreover, it inhibits pyruvate dehydrogenase complex both through feedback inhibition and phosphorylation through the activation of a specific kinase.
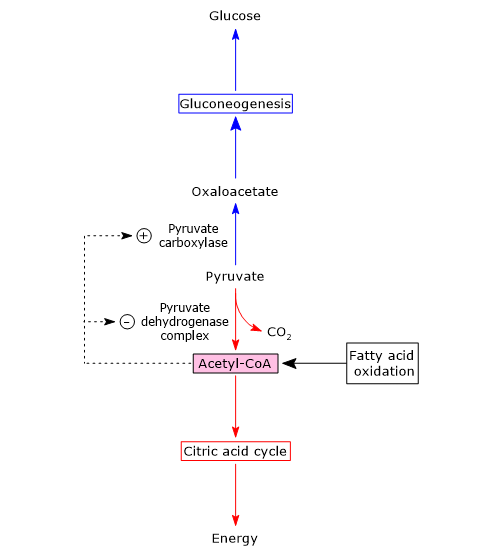
This means that when the energy charge of the cell is high, the formation of acetyl-CoA from pyruvate slows down, while the conversion of pyruvate to glucose is stimulated. Therefore acetyl-CoA is a molecule that signals that additional glucose oxidation for energy is not required and that glucogenic precursors can be used for the synthesis and storage of glucose.
Conversely, when acetyl-CoA levels decrease, the activity of pyruvate kinase and of the pyruvate dehydrogenase complex increases, and therefore also the flow of metabolites through the citric acid cycle. This supplies energy to the cell.
Summarizing, when the energy charge of the cell is high pyruvate carboxylase is active, and that the first control point of gluconeogenesis determines what will be the fate of pyruvate in the mitochondria.
Fructose 1,6-bisphosphatase
The second major control point in gluconeogenesis is the reaction catalyzed by fructose 1,6-bisphosphatase. The enzyme is allosterically inhibited by AMP. Therefore, when AMP levels are high, and consequently ATP levels are low, gluconeogenesis slows down. This means that, as previously seen, FBPase-1 is active when the energy charge of the cell is sufficiently high to support de novo synthesis of glucose.
Conversely, PFK-1, the corresponding glycolytic enzyme, is allosterically activated by AMP and ADP and allosterically inhibited by ATP and citrate, the latter resulting from the condensation of acetyl-CoA and oxaloacetate. Therefore:
- when AMP levels are high, gluconeogenesis slows down, and glycolysis accelerates;
- when ATP levels are high or when acetyl-CoA or citrate are present in adequate concentrations, gluconeogenesis is promoted, while glycolysis slows down.
The increase in citrate levels indicates that the activity of the citric acid cycle can slow down; in this way, pyruvate can be used in glucose synthesis.
PFK-1, FBPase-1 and fructose 2,6-bisphosphate
The liver plays a key role in maintaining blood glucose homeostasis: this requires regulatory mechanisms that coordinate glucose consumption and production. Two hormones are mainly involved: glucagon and insulin.[6]
They act intracellularly through fructose 2,6-bisphosphate or F2,6BP, an allosteric effector of PFK-1 and FBPase-1. This molecule is structurally related to fructose 1,6-bisphosphate, but is not an intermediate in glycolysis or gluconeogenesis.[1]
It was discovered in 1980 by Emile Van Schaftingen and Henri-Gery Hers, as a potent activator of PFK-1. In the subsequent year, the same researchers showed that it is also a potent inhibitor of FBPase-1.[9][10]
Fructose 2,6-bisphosphate, by binding to the allosteric site on PFK-1, reduces the affinity of the enzyme for ATP and citrate, allosteric inhibitors, and at the same time increases the affinity of the enzyme for fructose 6-phosphate, its substrate. PFK-1, in the absence of fructose 2,6-bisphosphate, and in the presence of physiological concentrations of ATP, fructose 6-phosphate, and of allosteric effectors AMP, ATP and citrate, is practically inactive. Conversely, the presence of fructose 2,6-bisphosphate activates PFK-1, thus stimulating glycolysis in the hepatocytes. At the same time fructose 2,6-bisphosphate slows down gluconeogenesis by inhibiting fructose 1,6-bisphosphatase, even in the absence of AMP. However, the effects of fructose-2,6-bisphosphate and AMP on FBPase-1 activity are synergistic.
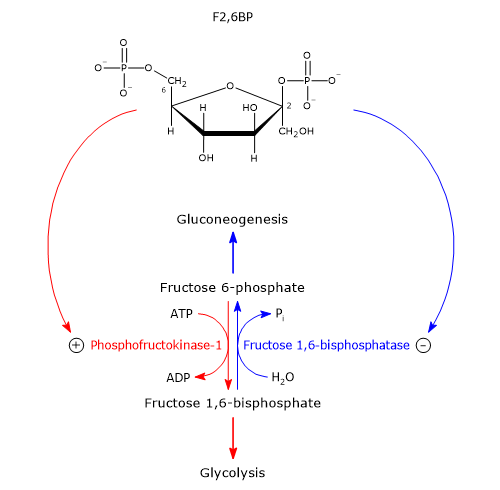
Fructose-2,6-bisphosphate concentration is regulated by the relative rates of synthesis and degradation. It is synthesized from fructose 6-phosphate in the reaction catalyzed by phosphofructokinase-2 or PFK-2 (EC 2.7.1.105), and is hydrolyzed to fructose 6-phosphate in the reaction catalyzed by fructose 2,6-bisphosphatase or FBPasi-2 (EC 3.1.3.46). These two enzymatic activities are located on a single bifunctional enzyme or tandem enzyme. In the liver, the balance of these two enzymatic activities is regulated by insulin and glucagon, as described below.
- Glucagon
It is released into the circulation when blood glucose levels drop, signaling the liver to reduce glucose consumption for its own needs and to increase de novo synthesis of glucose and its release from glycogen stores.
After binding to specific membrane receptors, glucagon stimulates hepatic adenylate cyclase (EC 4.6.1.1) to synthesize 3′,5′-cyclic AMP or cAMP, that activates cAMP-dependent protein kinase or protein kinase A or PKA (EC 2.7.11.11). The kinase catalyzes the phosphorylation, at the expense of one molecule of ATP, of a specific serine residue (Ser32) of PFK-2/FBPase-2. As a result of the phosphorylation, phosphatase activity increases while kinase activity decreases. Such reduction, due to the increase in the Km for fructose 6-phosphate, causes a decrease in the levels of fructose 2,6-bisphosphate, that, in turn, inhibits glycolysis and stimulates gluconeogenesis. Therefore, in response to glucagon, hepatic production of glucose increases, enabling the organ to counteract the fall in blood glucose levels.
Note: glucagon, like adrenaline, stimulates gluconeogenesis also by increasing the availability of substrates such as glycerol and amino acids. - Insulin
After binding to specific membrane receptors, insulin activates a protein phosphatase, the phosphoprotein phosphatase 2A or PP2A, that catalyzes the removal of the phosphate group from PFK-2/FBPase-2, thus increasing PFK-2 activity and decreasing FBPase-2 activity. (At the same time, insulin also stimulates a cAMP phosphodiesterase that hydrolyzes cAMP to AMP). This increases the level of fructose 2,6-bisphosphate, that, in turn, inhibits gluconeogenesis and stimulates glycolysis.
In addition, fructose 6-phosphate allosterically inhibits FBPase-2, and activates PFK-2. It should be noted that the activities of PFK-2 and FBPase-2 are inhibited by their reaction products. However, the main effectors are the level of fructose 6-phosphate and the phosphorylation state of the enzyme.
Glucose 6-phosphatase
Unlike pyruvate carboxylase and fructose-1,6-bisphosphatase, the catalytic subunit of glucose-6-phosphatase is not subject to allosteric or covalent regulation. The modulation of its activity occurs at the transcriptional level.[7] Low blood glucose levels and glucagon, namely, factors that lead to increased glucose production, and glucocorticoids stimulate its synthesis, that, conversely, is inhibited by insulin.
Also, the Km for glucose 6-phosphate is significantly higher than the range of physiological concentrations of glucose 6-phosphate itself. This is why it is said that the activity of the enzyme is almost linearly dependent on the concentration of the substrate, that is, enzyme is controlled by the level of substrate.
PEP carboxykinase
The enzyme is regulated mainly at the level of synthesis and degradation. For example, high levels of glucagon or fasting increase protein production through the stabilization of its mRNA and the increase in its transcription rate. High blood glucose levels or insulin have opposite effects.
Xylulose 5-phosphate
Xylulose 5-phosphate, a product of the pentose phosphate pathway, is a recently discovered regulatory molecule. It stimulates glycolysis and inhibits gluconeogenesis by controlling the levels of fructose 2,6-bisphosphate in the liver.
When blood glucose levels increase, e.g. after a meal high in carbohydrates, the activation of glycolysis and hexose monophosphate pathway occurs in the liver. Xylulose 5-phosphate produced activates protein phosphatase 2A, that, as previously said, dephosphorylates PFK-2/FBPase-2, thus inhibiting FBPase-2 and stimulating PFK-2. This leads to an increase in the concentration of fructose 2,6-bisphosphate, and then to the inhibition of gluconeogenesis and stimulation of glycolysis, resulting in increased production of acetyl-CoA, the main substrate for the synthesis of lipids.[2] At the same time, an increase in flow through the hexose monophosphate shunt occurs, leading to the production of NADPH, a source of electrons for lipid synthesis. Finally, PP2A also dephosphorylates carbohydrate-responsive element-binding protein or ChREBP, a transcription factor that activates the expression of hepatic genes for lipid synthesis. Therefore, in response to an increase in blood glucose levels, lipid synthesis is stimulated.
It is therefore evident that xylulose 5-phosphate is a key regulator of carbohydrate and fat metabolism.
Precursors
Besides the aforementioned pyruvate, the major gluconeogenic precursors are lactate, glycerol, the majority of the amino acids, and, more generally, any compound that can be converted to pyruvate or oxaloacetate.[1]
Glycerol
Glycerol is released by lipolysis in adipose tissue. With the exception of propionyl-CoA, it is the only part of the lipid molecule that can be used for de novo synthesis of glucose in animals.
Glycerol enters gluconeogenesis, or glycolysis, depending on the cellular energy charge, as dihydroxyacetone phosphate or DHAP, whose synthesis occurs in two steps.
In the first step, glycerol is phosphorylated to glycerol 3-phosphate, in the reaction catalyzed by glycerol kinase (EC 2.7.1.30), with the consumption of one ATP.
Glycerol + ATP → Glycerol 3-phosphate + ADP + Pi
The enzyme is absent in adipocytes but present in the liver; this means that glycerol needs to reach the liver to be further metabolized.
Glycerol 3-phosphate is then oxidized to dihydroxyacetone phosphate, in the reaction catalyzed by glycerol 3-phosphate dehydrogenase (EC 1.1.1.8). In this reaction NAD+ is reduced to NADH.
Glycerol 3-phosphate + NAD+ ⇄ Dihydroxyacetone phosphate + NADH + H+
During prolonged fasting, glycerol is the major gluconeogenic precursor, accounting for about 20 percent of glucose production.[3]
Glucogenic amino acids
Pyruvate and oxaloacetate are the entry points for the glucogenic amino acids, i.e. those whose carbon skeleton or part of it can be used for de novo synthesis of glucose.
Amino acids result from the catabolism of proteins, both food and endogenous proteins, like those of skeletal muscle during the fasting state or during intense and prolonged exercise.
The catabolic processes of each of the twenty amino acids that made up the proteins converge to form seven major products, acetyl-CoA, acetoacetyl-CoA, α-ketoglutarate, succinyl-CoA, fumarate, oxaloacetate, and pyruvate.
Except acetyl-CoA, acetoacetyl-CoA , the other five molecules can be used for gluconeogenesis. This means that gluconeogenic amino acids may also be defined as those whose carbon skeleton or part of it can be converted to one or more of the above molecules.
Below, the entry points of the gluconeogenic amino acids are shown.
- Pyruvate: alanine, cysteine, glycine, serine, threonine and tryptophan.
- Oxaloacetate: aspartate and asparagine.
- α-Ketoglutarate: glutamate, arginine, glutamine, histidine and proline.
- Succinyl-CoA: isoleucine, methionine, threonine and valine.
- Fumarate: phenylalanine and tyrosine.
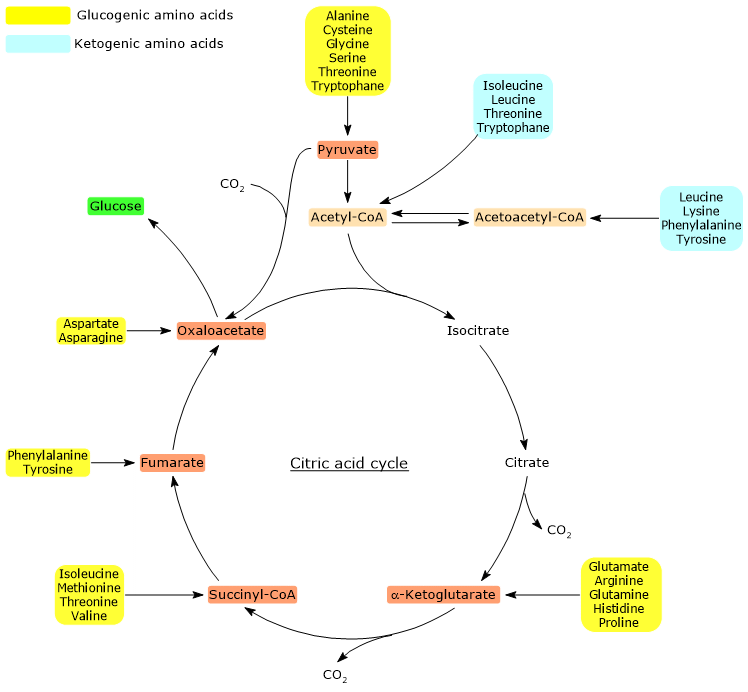
α-Ketoglutarate, succinyl-CoA and fumarate, intermediates of the citric acid cycle, enter the gluconeogenic pathway after conversion to oxaloacetate.
The utilization of the carbon skeletons of the amino acids requires the removal of the amino group. Alanine and glutamate, the key molecules in the transport of amino groups from extrahepatic tissues to the liver, are major glucogenic amino acids in mammals. Alanine is the main gluconeogenic substrate for the liver; this amino acid is shuttled to the liver from muscle and other peripheral tissues through the glucose-alanine cycle.
Ketogenic amino acids
Acetyl-CoA and acetoacetyl-CoA cannot be used for gluconeogenesis and are precursors of fatty acids and ketone bodies. The stoichiometry of the citric acid cycle elucidates why they cannot be used for de novo synthesis of glucose.
Acetyl-CoA, in the reaction catalyzed by citrate synthase, condenses with oxaloacetate to form citrate, a molecule with 6 carbon atoms instead of 4 as oxaloacetate. However, although the two carbon atoms from acetyl-CoA become part of the oxaloacetate molecule, two carbon atoms are oxidized and removed as CO2, in the reactions catalyzed by isocitrate dehydrogenase (EC 1.1.1.42) and α-ketoglutarate dehydrogenase complex. Therefore, acetyl-CoA does not yield any net carbon gain for the citric acid cycle.
Furthermore, the reaction leading to the formation of acetyl-CoA from pyruvate, catalyzed by the pyruvate dehydrogenase complex, that is the bridge between glycolysis and the Krebs cycle, is irreversible, and there is no other pathway to convert acetyl-CoA to pyruvate.
Pyruvate + NAD+ + CoASH → Acetyl-CoA + NADH + H+ + C02
For this reason, amino acids whose catabolism produces acetyl-CoA and/or acetoacetyl-CoA, are termed ketogenic.
Only leucine and lysine are exclusively ketogenic.
Note: Plants, yeasts, and many bacteria can use acetyl-CoA for de novo synthesis of glucose as they do have the glyoxylate cycle. This cycle has four reactions in common with the citric acid cycle, two unique enzymes, isocitrate lyase (EC 4.1.3.1) and malate synthase (EC 2.3.3.9), but lacks the decarboxylation reactions. Therefore, organisms that have such pathway are able to use fatty acids for gluconeogenesis.
Five amino acids, isoleucine, phenylalanine, tyrosine, threonine and tryptophan, are both glucogenic and ketogenic, because part of their carbon backbone can be used for gluconeogenesis, while the other gives rise to ketone bodies.
Propionate
Propionate, which belongs to the group of short-chain fatty acids, is a gluconeogenic precursor because, as propionyl-CoA, the active molecule, can be converted to succinyl-CoA.
Below, the different sources of propionate are analyzed.
- It may arise from beta-oxidation of odd-chain fatty acids such as margaric acid, a saturated fatty acid with 17 carbon atoms. Such fatty acids are rare compared to even-chain fatty acids, but present in significant amounts in the lipids of some marine organisms, ruminants, and plants. In the last pass through the β-oxidation sequence, the substrate is a five carbon fatty acid. This means that, once oxidized and cleaved to two fragments, it produces an acetyl-CoA and propionyl-CoA.
- Another source is the oxidation of branched-chain fatty acids, with alkyl branches with an odd number of carbon atoms. An example is phytanic acid, produced in ruminants by oxidation of phytol, a breakdown product of chlorophyll.
- In ruminants, propionate is also produced from glucose. Glucose is released from breakdown of cellulose by bacterial cellulase (EC 3.2.1.4) in the rumen, one of the four chambers that make up the stomach of these animals. These microorganisms then convert, through fermentation, glucose to propionate, which, once absorbed, may be used for gluconeogenesis, synthesis of fatty acids, or be oxidized for energy.[5]
In ruminants, in which gluconeogenesis tends to be a continuous process, propionate is the major gluconeogenic precursor. - Propionate may also result from the catabolism of valine, leucine, and isoleucine.
- In the colon, propionate is produced by bacteria of the gut microbiota through anaerobic fermentation of non-digestible carbohydrates, such as resistant starch and fibers. Absorbed by the enterocytes, the unmetabolized portion passes into the portal circulation and reaches the liver where it can enter gluconeogenesis.
The oxidation of propionyl-CoA to succinyl-CoA involves three reactions that occur in the liver and other tissues.
In the first reaction, propionyl-CoA is carboxylated to D-methylmalonyl-CoA in the reaction catalyzed by propionyl-CoA carboxylase (EC 6.4.1.3), a biotin-requiring enzyme. This reaction consumes one ATP.
Propionyl-CoA + HCO3– + ATP → D-methylmalonyl-CoA+ ADP + Pi
In the subsequent reaction, catalyzed by methylmalonyl-CoA epimerase (EC 5.1.99.1), D-methylmalonyl-CoA is epimerized to its L-stereoisomer.
D-Methylmalonyl-CoA ⇄ L-Methylmalonyl-CoA
Finally, L-methylmalonyl-CoA undergoes an intramolecular rearrangement to succinyl-CoA, in the reaction catalyzed by methylmalonyl-CoA mutase (EC 5.4.99.2). This enzyme requires 5-deoxyadenosylcobalamin or coenzyme B12, a derivative of cobalamin or vitamin B12, as a coenzyme.
L-Methylmalonyl-CoA ⇄ Succinyl-CoA
References
- ^ a b c d e f g Garrett R.H., Grisham C.M. Biochemistry. 4th Edition. Brooks/Cole, Cengage Learning, 2010
- ^ Kabashima T., Kawaguchi T., Wadzinski B.E., Uyeda K. Xylulose 5-phosphate mediates glucose-induced lipogenesis by xylulose 5-phosphate-activated protein phosphatase in rat liver. Proc Natl Acad Sci USA 2003;100:5107-5112. doi:10.1073/pnas.0730817100
- ^ Kuriyama H. et all. Coordinated regulation of fat-specific and liver-specific glycerol channels, aquaporin adipose and aquaporin 9. Diabetes 2002;51(10):2915-2921. 10.2337/diabetes.51.10.2915
- ^ McCommis K.S. and Finck B.N. Mitochondrial pyruvate transport: a historical perspective and future research directions. Biochem J 2015;466(3):443-454. doi:10.1042/BJ20141171
- ^ a b c d e f g h i Nelson D.L., M. M. Cox M.M. Lehninger. Principles of biochemistry. 6th Edition. W.H. Freeman and Company, 2012
- ^ a b c d e Rosenthal M.D., Glew R.H. Medical biochemistry – Human metabolism in health and disease. John Wiley J. & Sons, Inc., Publication, 2009
- ^ Soty M., Chilloux J., Delalande F., Zitoun C., Bertile F., Mithieux G., and Gautier-Stein A. Post-Translational regulation of the glucose-6-phosphatase complex by cyclic adenosine monophosphate is a crucial determinant of endogenous glucose production and is controlled by the glucose-6-phosphate transporter. J Proteome Res 2016;15(4):1342-1349. doi:10.1021/acs.jproteome.6b00110
- ^ a b c Stipanuk M.H., Caudill M.A. Biochemical, physiological, and molecular aspects of human nutrition. 3rd Edition. Elsevier health sciences, 2012
- ^ Van Schaftingen E., and Hers H-G. Inhibition of fructose-1,6-bisphosphatase by fructose-2,6-bisphosphate. Proc Natl Acad Sci USA 1981;78(5):2861-2863. doi:10.1073/pnas.78.5.2861
- ^ Van Schaftingen E., Jett M-F., Hue L., and Hers H-G. Control of liver 6-phosphofructokinase by fructose 2,6-bisphosphate and other effectors. Proc Natl Acad Sci USA 1981;78(6):3483-3486. doi:10.1073/pnas.78.6.3483