Proanthocyanidins or condensed tannins, also called pycnogenols and leukocyanidins, are polyphenolic compounds (in particular they are a flavonoid subgroup) widely distributed in the plant kingdom, second only to lignin as the most abundant phenol in nature.
They are present in high concentrations in various parts of the plants such as flowers, fruits, berries, seeds (e.g. in grape seeds), and bark (e.g. pine bark).
Together with anthocyanins and their oxidation products, and catechins, they are the most abundant flavonoids in human diet and it has been suggested that they constitute a significant fraction of the polyphenols ingested in the Western diet.
Therefore, condensed tannins should be taken into consideration when the epidemiological association between the intake of polyphenols, especially flavonoids, and chronic diseases are examined.
Contents
Chemical structure
Condensed tannins have a complex chemical structure being oligomers (dimers to pentamers) or polymers (six or more units, up to 60) of catechins or flavanols, which are joined by carbon-carbon bonds.
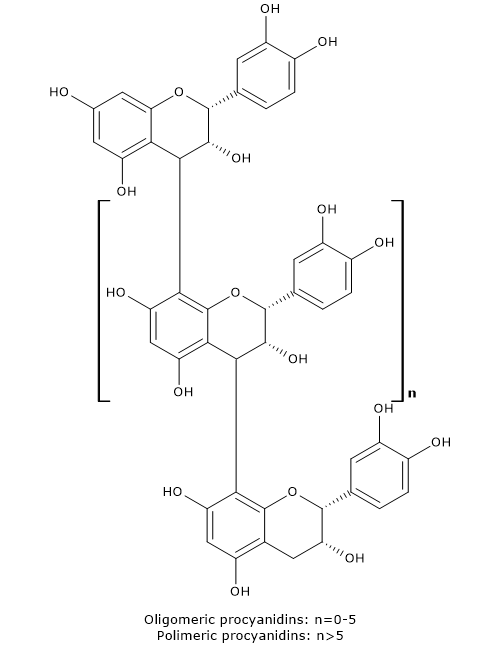
They may consist exclusively of:
- (epi)catechin, and they are named procyanidins;
- (epi)afzelechin, and they are named propelargonidins;
- (epi)gallocatechin, and they are named prodelphinidins.
Propelargonidins and prodelphinidins are less common in nature and in foods than procyanidins.
Depending on the bonds between monomers, proanthocyanidins have a:
- B-type structure, if the polymerization occurs via carbon-carbon bond between the position 8 of the terminal unit and the 4 of the extender (or C4-C6);
- A-type structure, less frequent, if monomers are doubly linked via an ether bond C2-O-C7 or C2-O-C5 plus a B-type bond.
Procyanidins
The most common dimers are B-type procyanidins, B1 to B8, formed by catechin or epicatechin; in B1, B2, B3 and B-4 dimers, the two flavanol units are joined by a C4-C8 bond; in B5, B6, B7 and B8 dimers the two units are joined by C4-C6 bond.
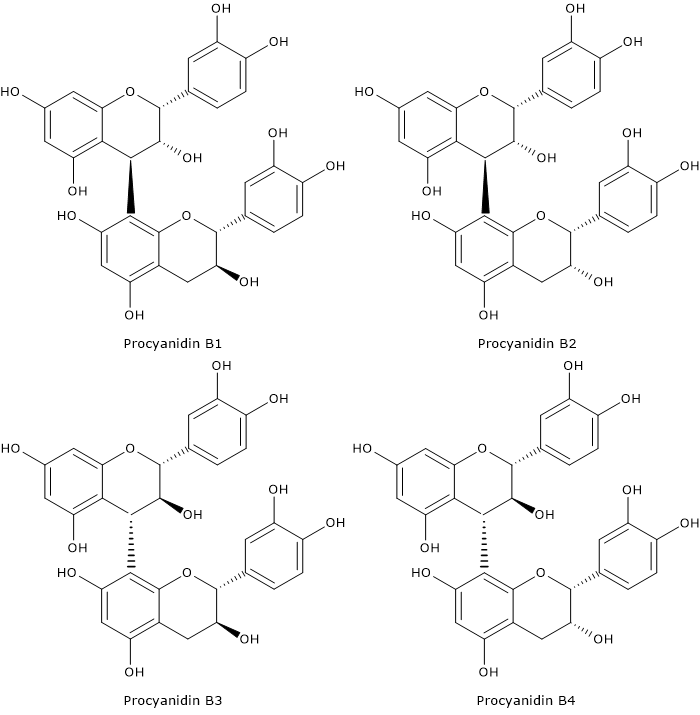
Procyanidin C1 is a B-type trimer.
Procyanidin A-2 is an example of A-type procyanidin.
Intestinal absorption
Condensed tannins are poorly absorbed from the intestine; together with anthocyanins and gallic acid ester derivatives of tea catechins, they are the least well-absorbed polyphenols.
It seems that low molecular weight oligomers (2-3 monomers) may be absorbed as such while polymers are not.
In the systemic circulation, dimers reach concentrations of two orders of magnitude lower than those of catechins.
It seems that condensed tannins with a degree of polymerization greater than three transit into the stomach and small intestine without significant modifications, and then, into the large intestine, they are catabolized by gut microbiota, which is part of the human microbiota, with production of phenylpropionic, phenilvaleric and phenylacetic acids. These degradation products have been suggested to be the major metabolites of proanthocyanidins in healthy humans.
Procyanidins and catechins
It had been proposed that the catabolism of procyanidins in the gastrointestinal tract lead to the release of monomeric catechins, thus indirectly contributing to their systemic pool in humans. In recent years, it has been shown that this does not happen because procyanidins do not significantly contribute to:
- the concentration of catechin metabolites in the systemic circulation;
- the total catechin metabolites excreted in the urine;
- finally, they do not significantly affect plasma metabolite profile derived from catechol-O-methyltransferase activity.
Therefore, analyzing the potential health benefits associated with the intake of foods containing these phytochemicals, catechins and procyanidins should be considered distinct classes of related compounds.
References
- de la Rosa L.A., Alvarez-Parrilla E., Gonzàlez-Aguilar G.A. Fruit and vegetable phytochemicals: chemistry, nutritional value, and stability. 1th Edition. Wiley J. & Sons, Inc., Publication, 2010
- Gu L., Kelm M.A., Hammerstone J.F., Beecher G., Holden J., Haytowitz D., Gebhardt S., and Prior R.L. Concentrations of proanthocyanidins in common foods and estimations of normal consumption. J Nutr 2004;134(3):613-617. doi:10.1093/jn/134.3.613
- Han X., Shen T. and Lou H. Dietary polyphenols and their biological significance. Int J Mol Sci 2007;9:950-988. doi:10.3390/i8090950
- Manach C., Scalbert A., Morand C., Rémésy C., and Jime´nez L. Polyphenols: food sources and bioavailability. Am J Clin Nutr 2004;79(5):727-747. doi:10.1093/ajcn/79.5.727
- Nandakumar V., Singh T., and Katiyar S.K. Multi-targeted prevention and therapy of cancer by proanthocyanidins. Cancer Lett 2008;269(2):378-387. doi:10.1016/j.canlet.2008.03.049
- Ottaviani J.I., Kwik-Uribe C., Keen C.L., and Schroeter H. Intake of dietary procyanidins does not contribute to the pool of circulating flavanols in humans. Am J Clin Nutr 2012;95:851-858. doi:10.3945/ajcn.111.028340
- Santos-Buelga C. and Scalbert A. Proanthocyanidins and tannin-like compounds: nature, occurrence, dietary intake and effects on nutrition and health. J Sci Food Agr 2000;80(7):1094-1117. doi:10.1002/(SICI)1097-0010(20000515)80:7<1094::AID-JSFA569>3.0.CO;2-1
- Tsao R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010;2:1231-1246. doi:10.3390/nu2121231
- Wang Y.,Chung S., Song W.O., and Chun O.K. Estimation of daily proanthocyanidin intake and major food sources in the U.S. diet. J Nutr 2011;141(3):447-452. doi:10.3945/jn.110.133900